Tag: pharmaceutical supply chain
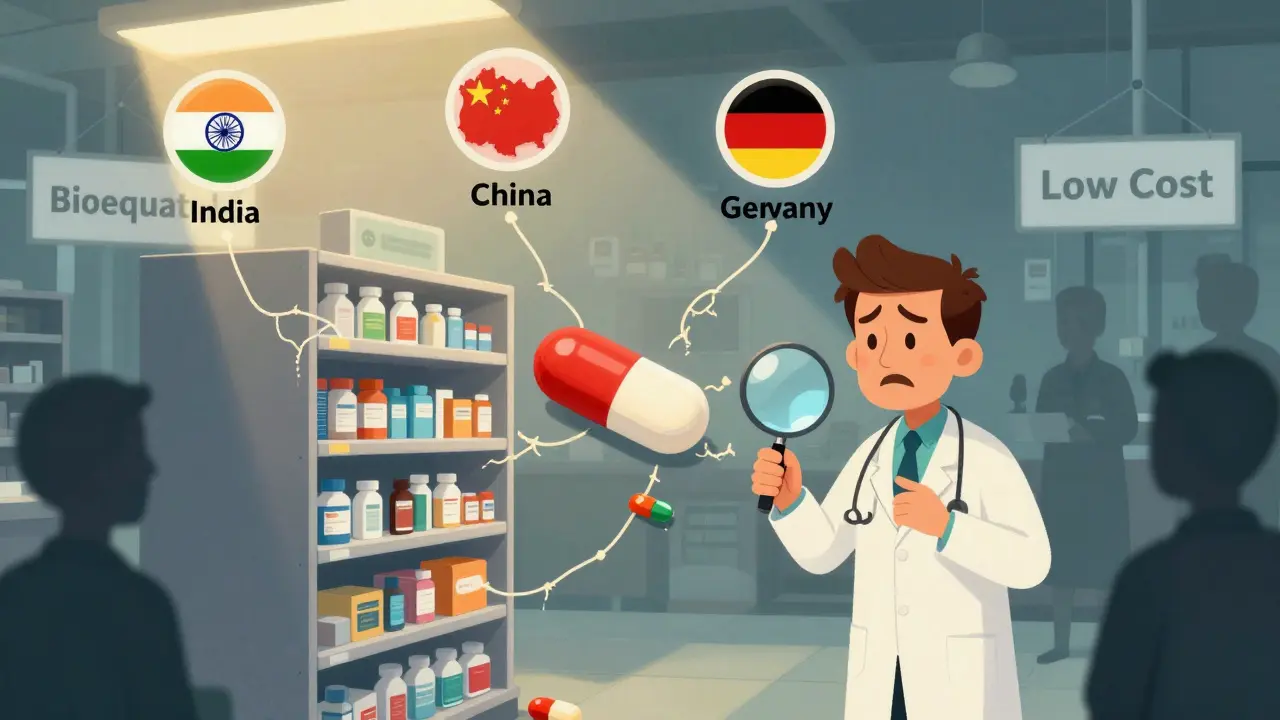
Quality concerns: when clinicians question generic manufacturing
Clinicians are raising alarms about generic drug quality as manufacturing shifts overseas. Studies show higher adverse event rates for drugs made in India, and outdated supply chains are causing dangerous shortages. Transparency and domestic production may be the key to safer generics.
read morerecent posts

Confused by the dates on your medicine bottle? Learn the critical difference between Expiration Dates and Refill-By Dates to ensure safety and avoid treatment gaps.
Published ON: 25 Mar
Explore how the TRIPS agreement shapes generic medicine access globally. Learn about patent laws, compulsory licensing, and the balance between innovation and public health.
Published ON: 30 Mar
Contamination controls in generic drug manufacturing prevent dangerous cross-contamination and ensure patient safety. Learn how cleanrooms, real-time monitoring, and human protocols keep generics safe-and why regulators are cracking down.
Published ON: 8 Mar
Managing therapeutic equivalence in combination drugs requires more than matching active ingredients. Dose interactions, formulation differences, and NTI risks can lead to serious errors-even when generics are FDA-approved.
Published ON: 21 Mar
Switching health plans? Your generic drug costs could spike if you don't check formulary tiers. Learn how tier structures, deductibles, and state rules affect what you pay-and how to avoid costly surprises.
Published ON: 14 Mar