Tag: generic manufacturing
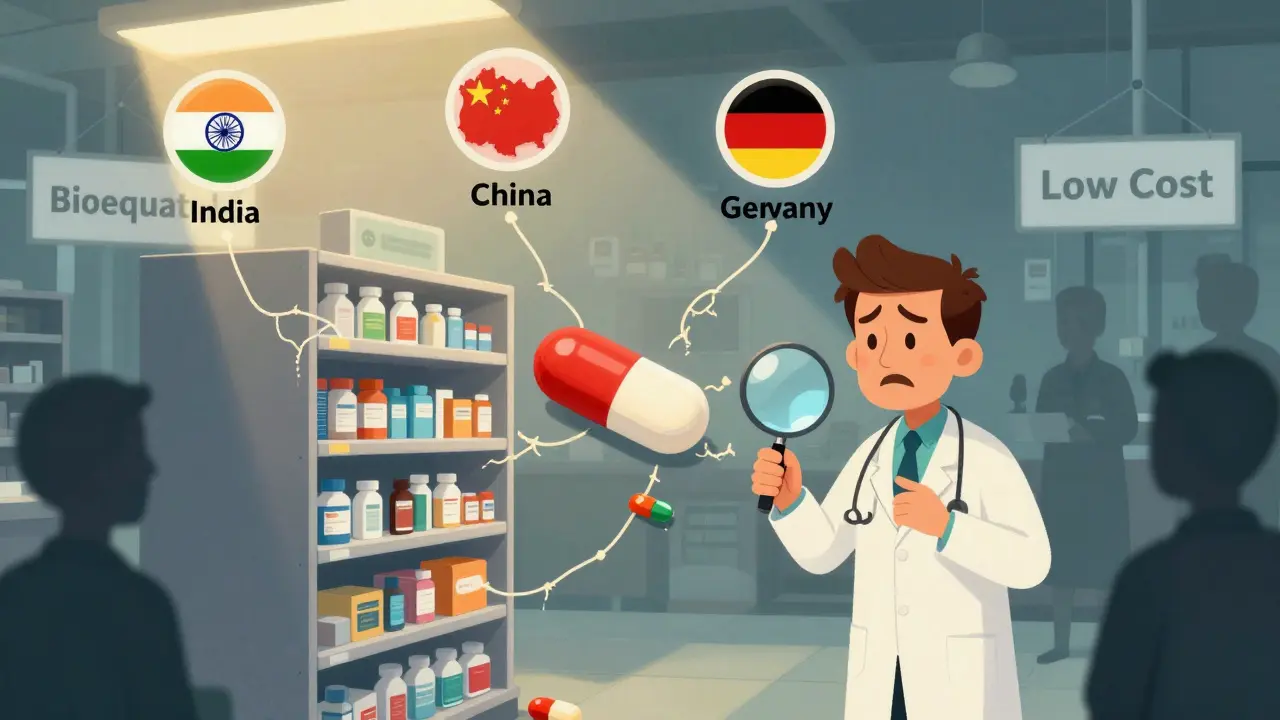
Quality concerns: when clinicians question generic manufacturing
Clinicians are raising alarms about generic drug quality as manufacturing shifts overseas. Studies show higher adverse event rates for drugs made in India, and outdated supply chains are causing dangerous shortages. Transparency and domestic production may be the key to safer generics.
read morerecent posts
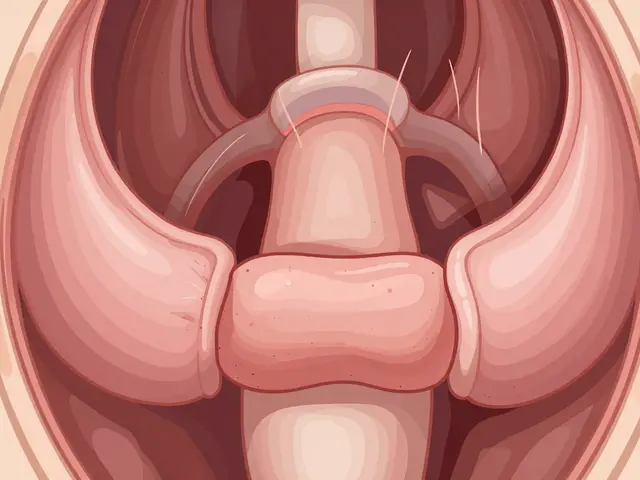
Learn how esophageal motility disorders affect swallowing and digestion. Discover the role of high-resolution manometry and the Chicago Classification in diagnaching dysphagia and treating conditions like achalasia.
Published ON: 29 Mar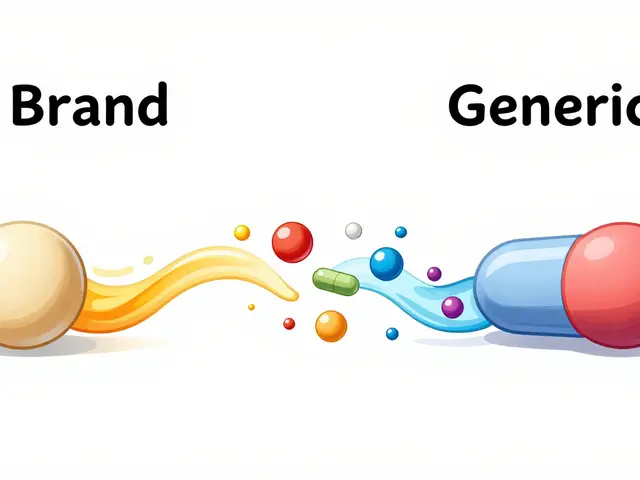
Managing therapeutic equivalence in combination drugs requires more than matching active ingredients. Dose interactions, formulation differences, and NTI risks can lead to serious errors-even when generics are FDA-approved.
Published ON: 21 Mar
Explore how the TRIPS agreement shapes generic medicine access globally. Learn about patent laws, compulsory licensing, and the balance between innovation and public health.
Published ON: 30 MarMeasuring patient education effectiveness isn’t about surveys or handouts-it’s about seeing if patients can truly apply what they’ve learned. Learn how to track real understanding using simple, evidence-based methods that work in clinics today.
Published ON: 7 Mar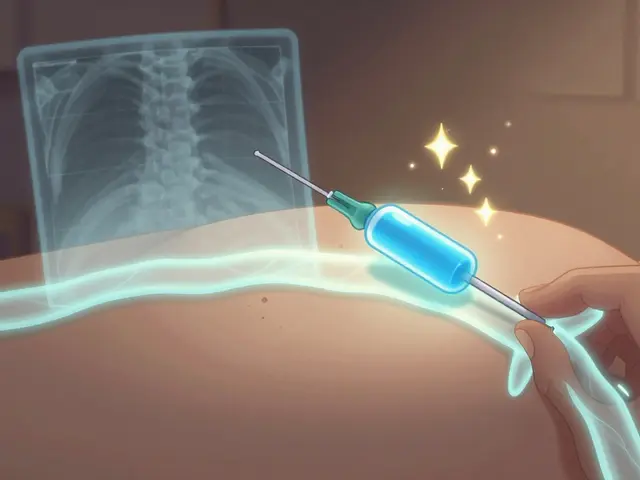
Nerve blocks and radiofrequency ablation (RFA) are key interventional pain procedures that offer long-term relief for chronic pain. Learn how they work, who they help, and why RFA is becoming a standard treatment for back, knee, and neck pain.
Published ON: 18 Mar